Mean Free Path, Molecular Collisions
$ 10.99 · 4.8 (651) · In stock
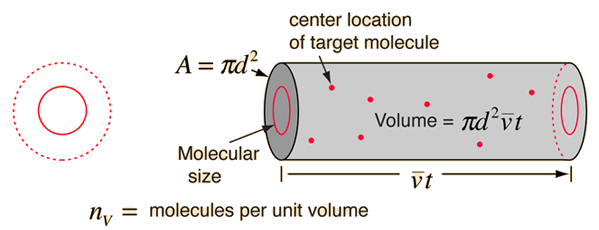

The mean path lambda of molecules is given by where n is the number of molecules per unit volume and d is the diameter of the molecules.
20. The mean free path of benzene molecule is6 x 10 6 m and average speed of molecules is1.2 x 103 m/s. The collision rate of molecules, is(1) 3 x 107 s

What is the significance of a mean free path? - Quora

thermodynamics - Mean free path of molecules - Physics Stack Exchange

Mean Free Path, Molecular Collisions
Mean Free Path, PDF, Gases

Mean free path - Wikipedia

Why in gases, interatomic distance is of order of tens of A° but mean free path of 1000s of A°? - Quora

Mean Free Path, Molecular Collisions
