Modular Medical submits next-gen insulin pump for FDA clearance
$ 19.99 · 5 (248) · In stock

Modular Medical (Nasdaq:MODD) announced today that it submitted its next-generation MODD1 insulin pump to the FDA for 510(k) clearance.
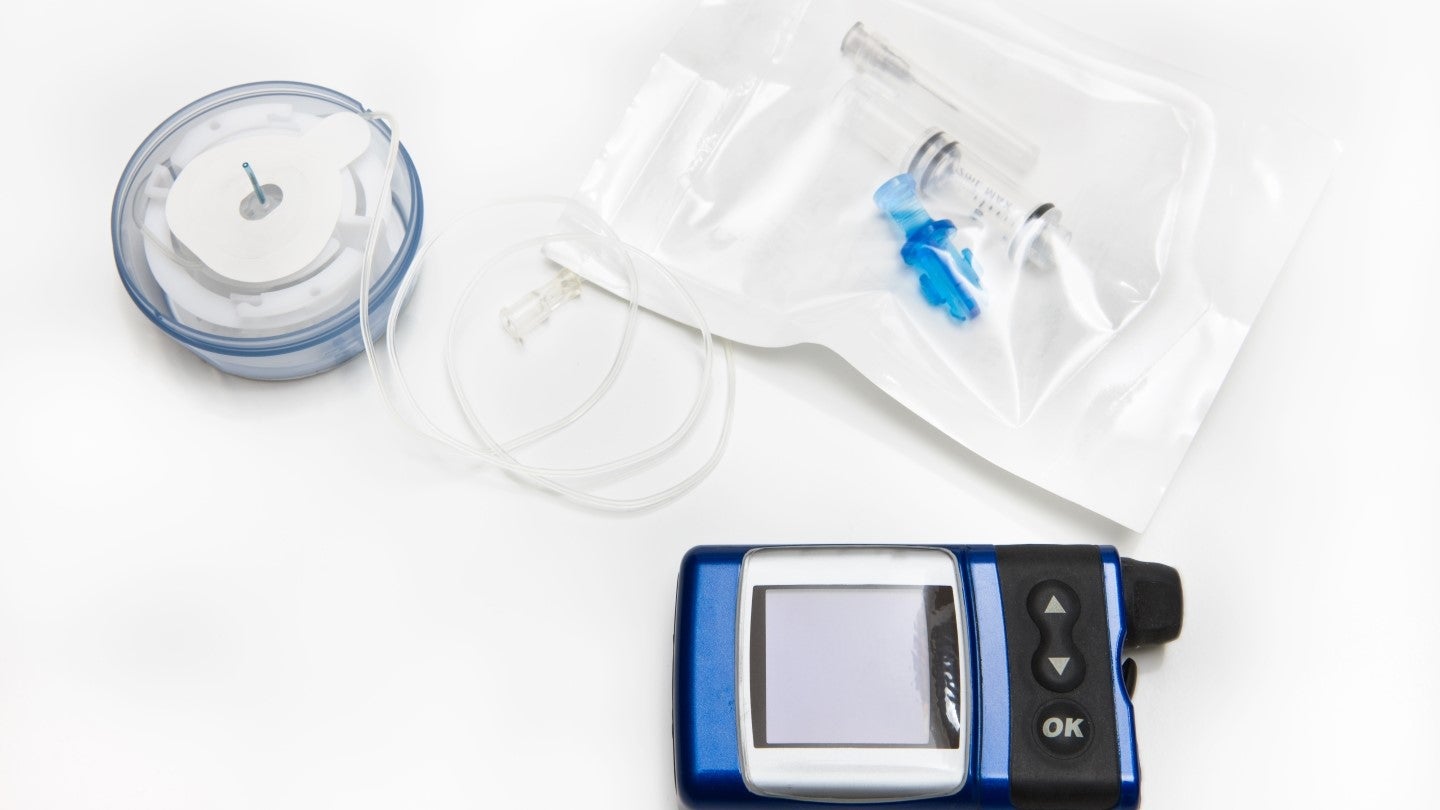
PharmaSens seeks FDA approval for new insulin pump
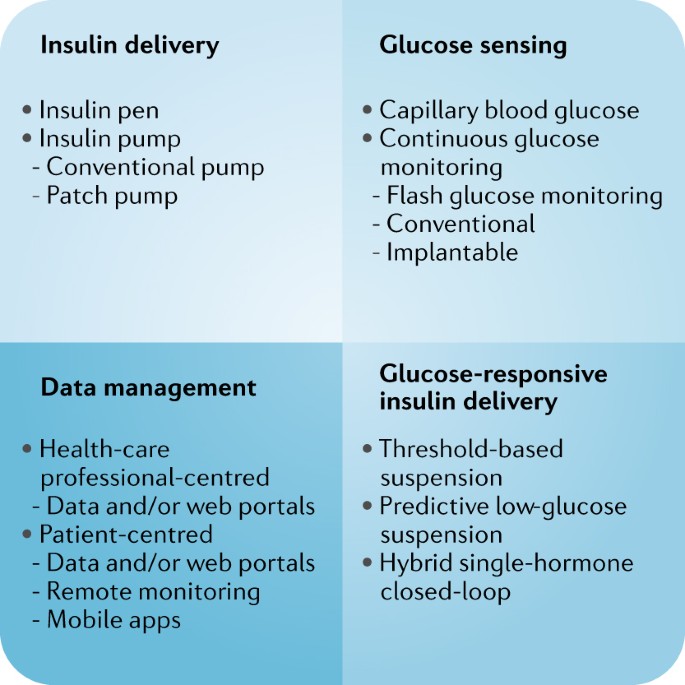
Technology in the management of type 1 diabetes mellitus — current status and future prospects

The stem cell 'skin gun' that's aiming to disrupt wound care - MassDevice

FDA roundup: The major device, app, and algorithm approvals of 2018 (so far)

Medical devices, smart drug delivery, wearables and technology for the treatment of Diabetes Mellitus - ScienceDirect
Drug Delivery Business News on LinkedIn: Nemaura Medical garners U.S. purchase order for glucose sensors

Medtronic launches smartphone connectivity for CGMs, insulin pumps

Automated insulin delivery: benefits, challenges, and recommendations. A Consensus Report of the Joint Diabetes Technology Working Group of the European Association for the Study of Diabetes and the American Diabetes Association
David Kliff en LinkedIn: Dexcom to announce U.S. G7 launch in Super Bowl commercial with Nick Jonas

Upgraded Sapphire Infusion Pump System Receives FDA Clearance - OR Today

Novocure touts combined TTF, chemotherapy mesothelioma study data - MassDevice
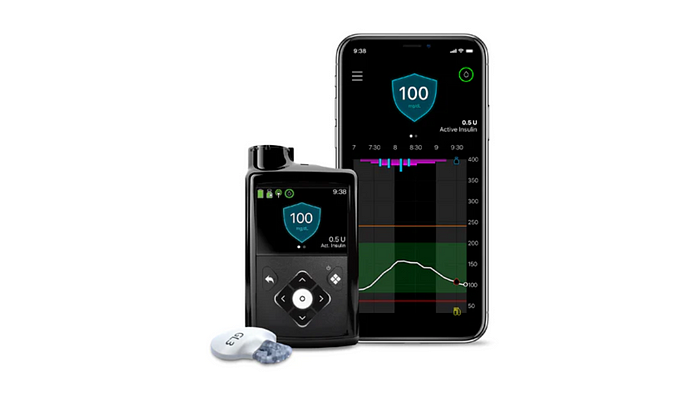
Why people with type 1 diabetes should (not) avoid these Top 10 AID Systems
Erica Scott - CSAM on LinkedIn: Modular Medical submits next-gen insulin pump for FDA clearance

In the News.. weight loss & cancer study for T2D, new pump submitted, Summer Olympic hopeful with T1D and more! - Diabetes Connections


