My publications - CHM 201-LECTURE IV-REAL GASES - Page 8 - Created with Publitas.com
$ 18.99 · 4.5 (539) · In stock
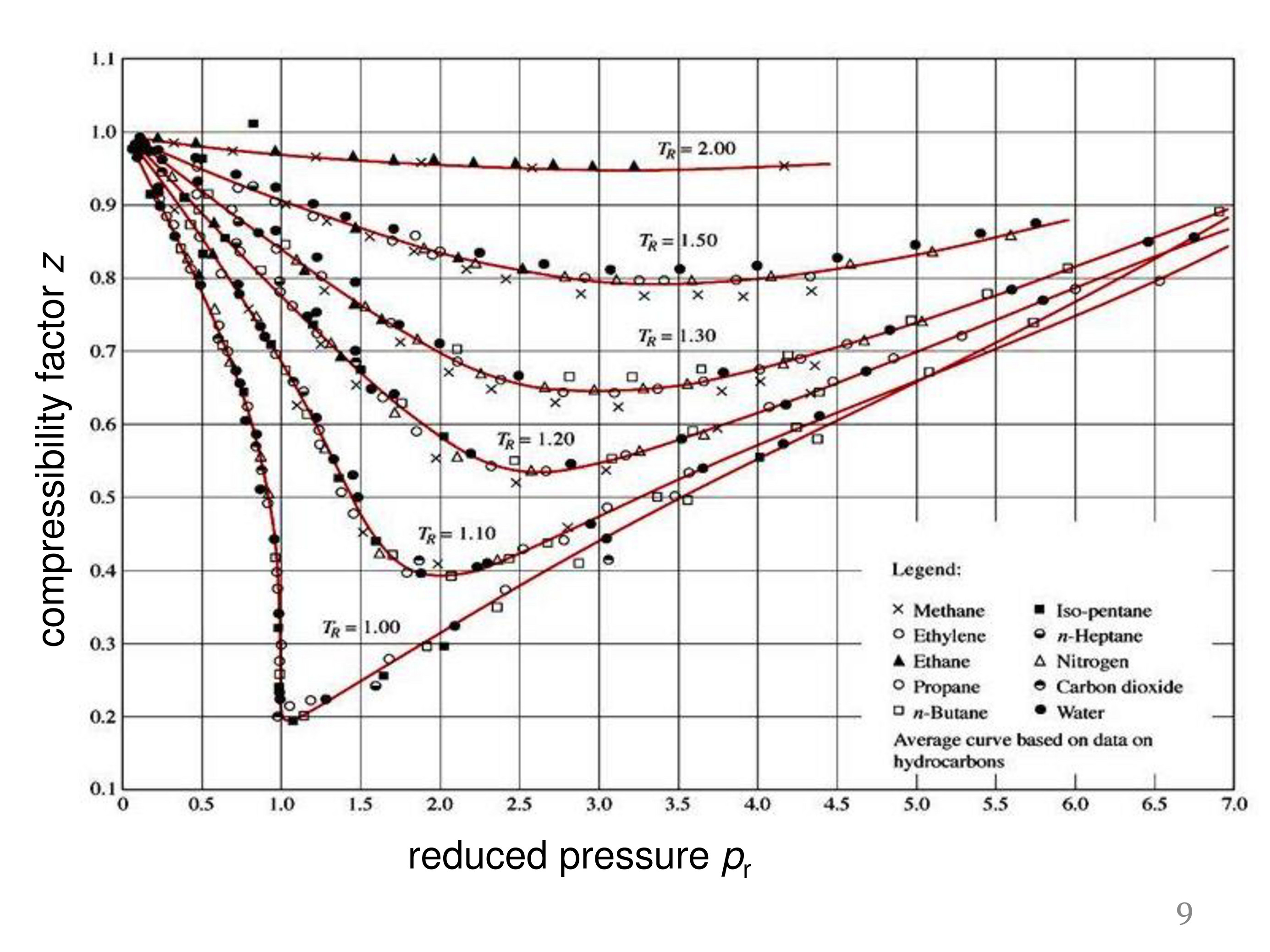
• Compressibility factor z: ▪ Dimensionless quantity. ▪ For a pure substance, it is a function of temperature and pressure or temperature and molar volume. ▪
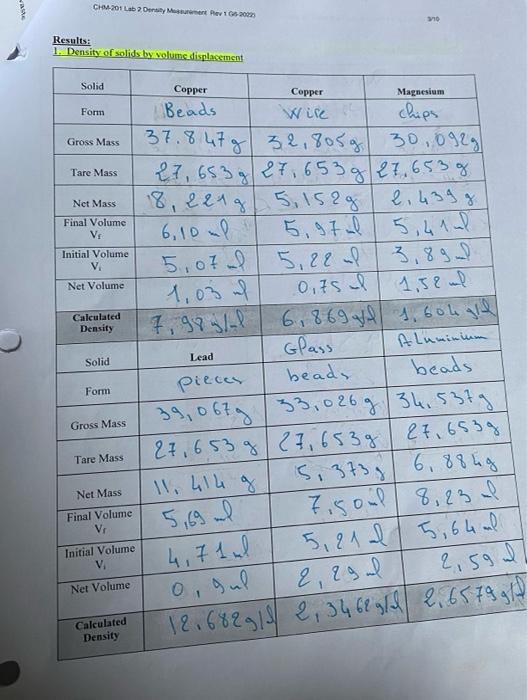
Solved CHM-201 General Chemistry and Laboratery I Laboratory

My publications - CHM 201-LECTURE IV-REAL GASES - Page 8 - Created

SOLUTION: Gaseous state practice sheet gaseous state chemistry

CHEM 201] - Final Exam Guide - Ultimate 59 pages long Study Guide! - OneClass

My publications - CHM 201-LECTURE II-CHEMICAL THERMODYNAMICS

Science

CHM 2210L : Organic Chemistry 1 Lab - USF

Solved Styles Editing Voice CHEM 1411 Determination of R
Solved CHM-201 General Chemistry and Laboratery I Laboratory