Derive an expression for the compression factor of a gas tha
$ 19.99 · 4.9 (337) · In stock
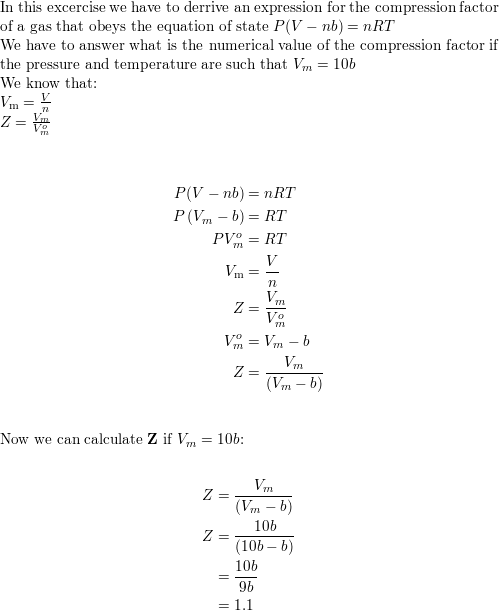

b 26. The compressibility factor 1 mole of a van der Waal's gas Boyle temperature is 1+ VIV-yo) Find the value of x + y. tronarding the van property?

Physical Chemistry Atkins 10th homework all - ####### 54 1 The properties of gases CHAPTER 1 The - Studocu

Answered: 16. The PVT data as shown below were…

Real Gases and the Virial Equation
Compressibility factor (gases) - Knowino

Solved 1, A gas at 250 K and 1 atm has a molar volume 5%
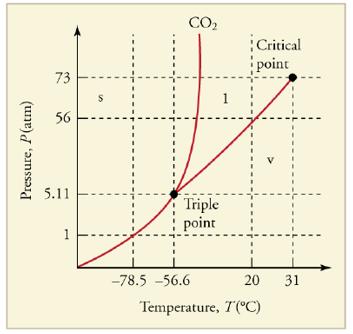
What is the vapor pressure of solid carbon dioxide (dry ice) at _785℃ Figure 13.36 The phase diagram for carbon dioxide. The axes aha nonlinear, and the graph is not to scale.
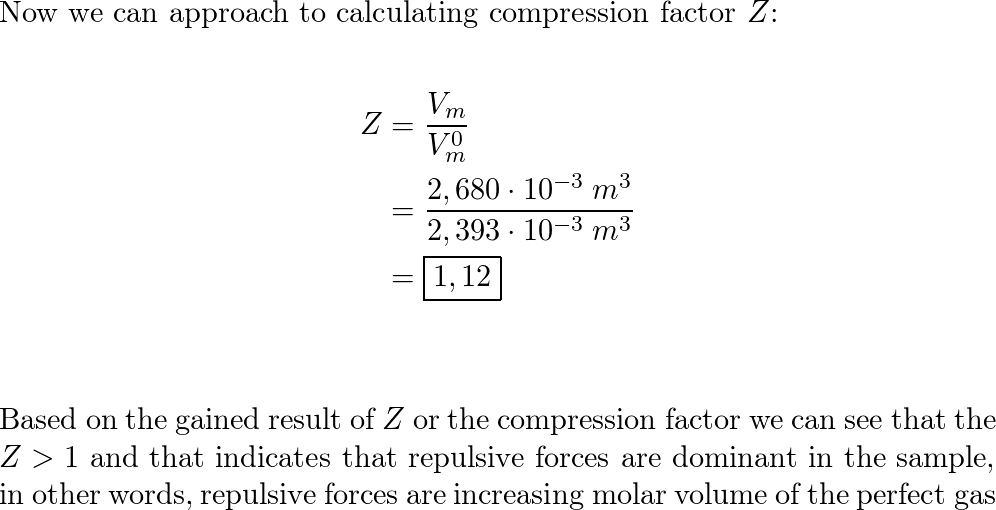
a) A gas at 250 K and 15 atm has a molar volume 12 per cent

Material Balance Analysis Theory

Atkins' Physical Chemistry [11th ed.] 0198769865, 9780198769866